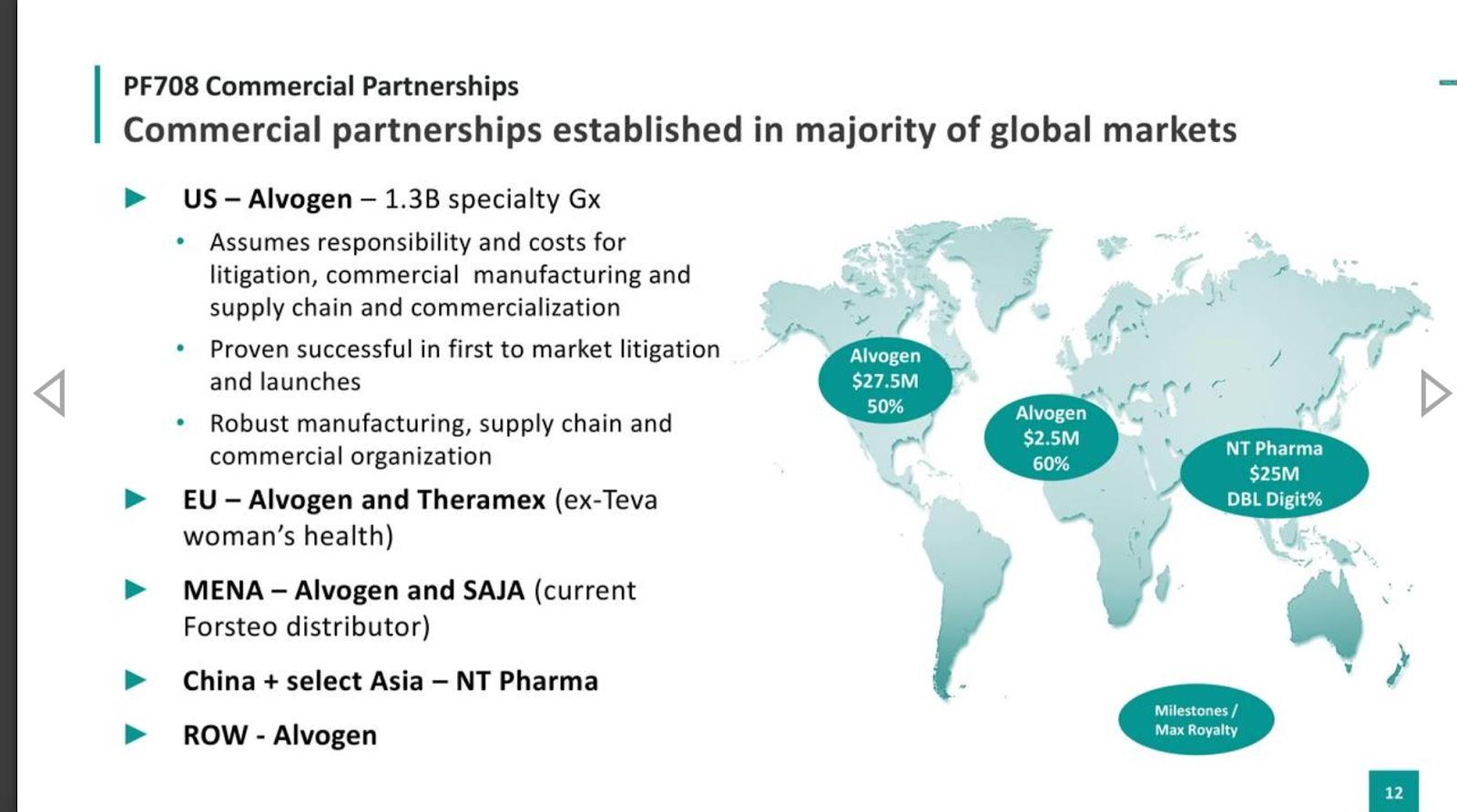
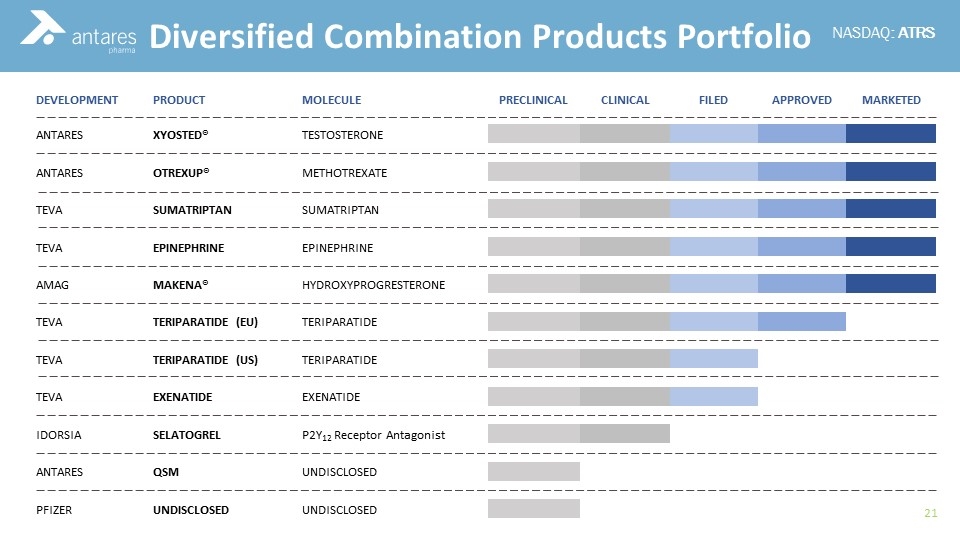
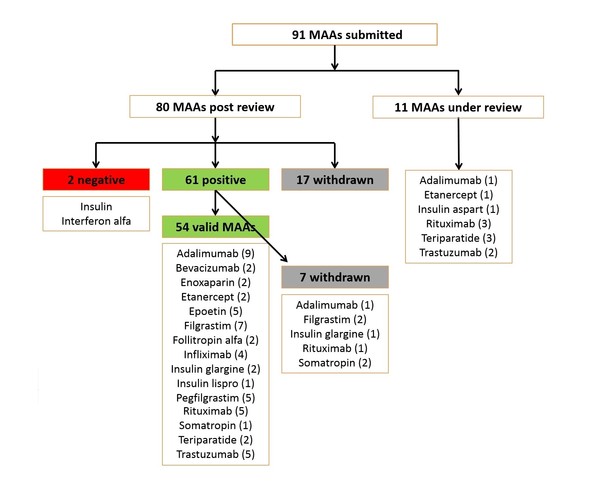
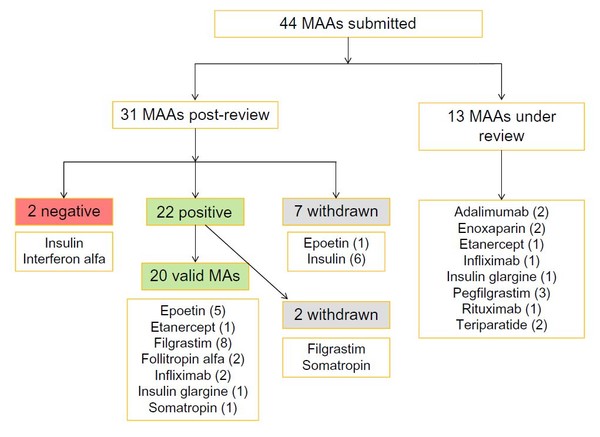
Teva launch Tetridar® (teriparatide 20 microgram/80 microlitres Solution for injection), a pre-filled pen injection for the treatment of osteoporosis. - IPN Beta

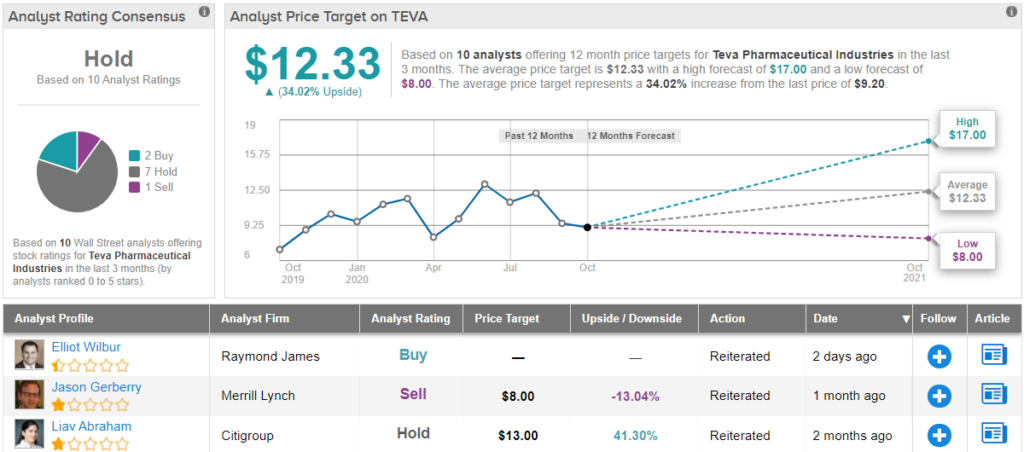
Pfenex: The First U.S. Forteo Biosim And A Host Of Other Biosims In Pipeline (NYSEMKT:PFNX) | Seeking Alpha

Obvious to the Bone: Teva Wins Opposition against Eli Lilly's Patent Application Covering Osteoporosis Blockbuster FORTEO® - Lexology